Trending...
- Platinum Car Audio LLC Focuses on Customer-Driven Vehicle Audio and Electronics Solutions
- Mecpow M1: A Safe & Affordable Laser Engraver Built for Home DIY Beginners
- Postmortem Pathology Expands Independent Autopsy Services in Kansas City
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) $NRXP; $750 Million Market for Suicidal Depression Market Awaits
MIAMI - PennZone -- A $750 million global market for preservative-free IV ketamine is now within reach for NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP), following the company's announcement that the U.S. Food and Drug Administration (FDA) has formally accepted its Abbreviated New Drug Application (ANDA) for KETAFREE™, the first known preservative-free ketamine formulation. The FDA has deemed the application "substantially complete," assigning a GDUFA target action date of July 29, 2026—a critical milestone that positions NRx for potential entry into a high-demand therapeutic market.
The FDA submission comes as independent analyst D. Boral issues a Buy rating with a $34 price target, citing NRx's maturing regulatory position, expanding revenue-generating operations, and strong clinical catalysts across its NMDA-focused drug platform.
A Transformative Solution for a Critical Mental Health Crisis
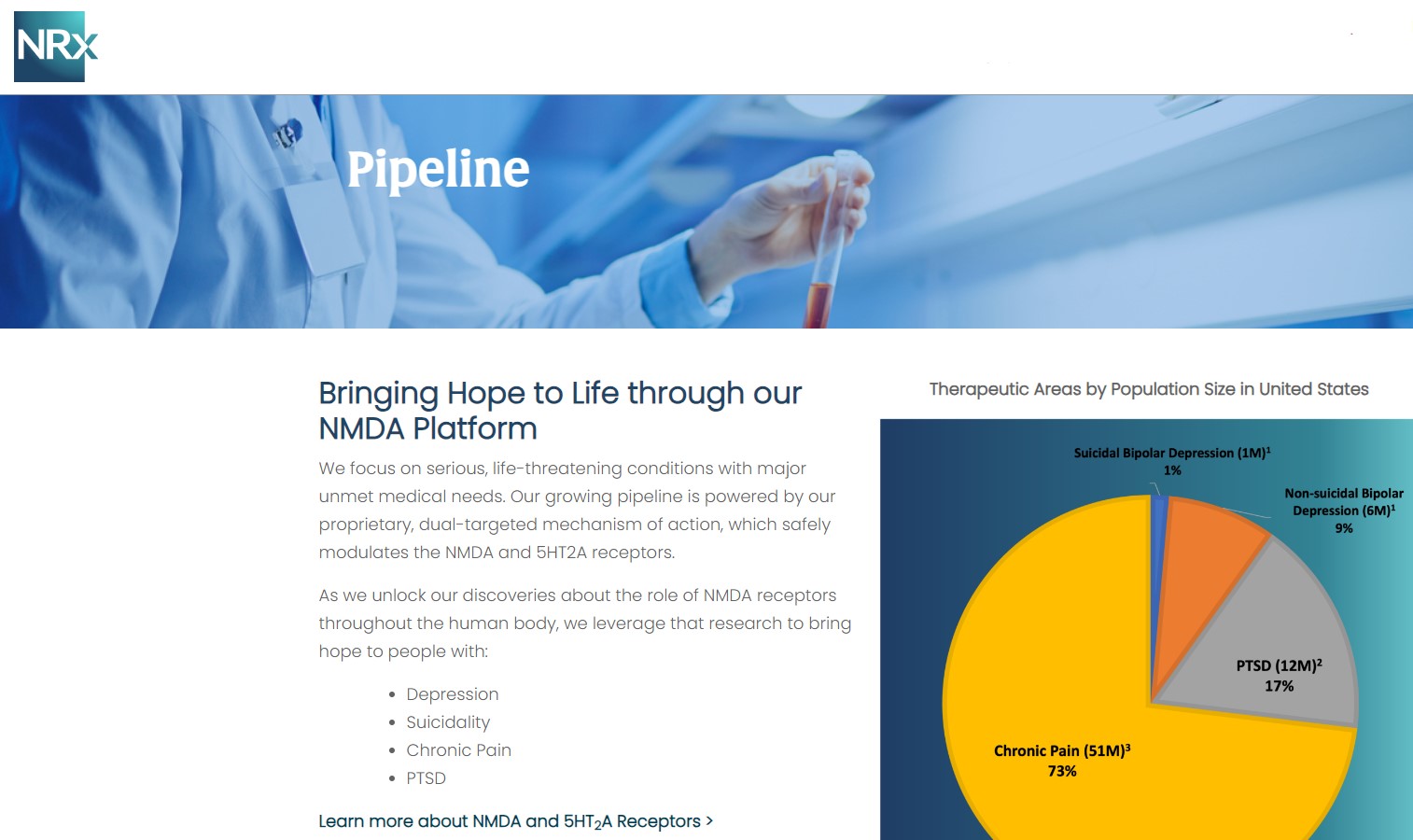
More than 13 million Americans contemplate suicide each year, according to the CDC. Current treatment options are limited, and many lack evidence of rapid antisuicidal benefit. NRx's pipeline directly targets this gap:
NRX-100 (IV Ketamine)
NRX-101 (D-Cycloserine + Lurasidone)
KETAFREE™: A Strategic ANDA Pathway and Major Competitive Advantage
More on The PennZone
The global generic ketamine market is estimated at $750 million annually. Nearly all existing products contain benzethonium chloride (BZT)—a preservative banned from topical antiseptics and not recognized as safe by the FDA.
NRx's preservative-free KETAFREE™ directly addresses this safety concern.
The company has:
KETAFREE™ is strategically separate from the company's innovative NRX-100 program, enabling two simultaneous FDA pathways:
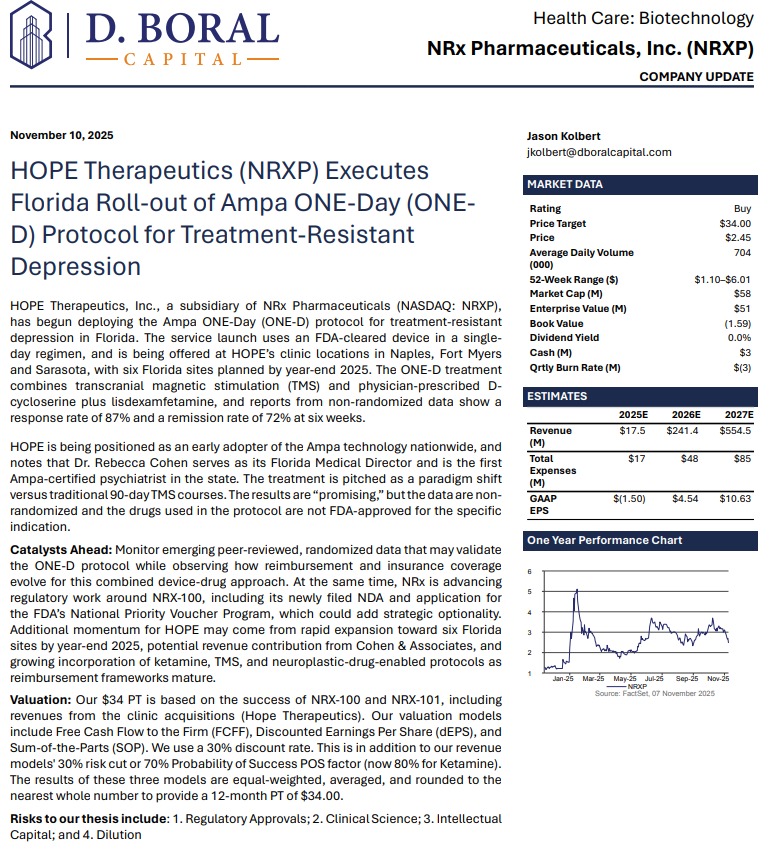
Expanding Commercial Footprint: HOPE Therapeutics Clinics
2025 marks NRx's entry into revenue-generating operations through its HOPE Therapeutics subsidiary.
The company currently operates three clinics in Florida, with six more expected by year-end, targeting treatment-resistant psychiatric conditions, chronic pain, and military/veteran mental health.
NRx also launched Florida's first deployment of the ONE-D protocol with Ampa Health, a groundbreaking, single-day treatment approach utilizing an FDA-cleared device. Peer-reviewed results of the protocol show up to 87% response and 72% remission rates, signaling a disruptive new pathway for addressing treatment-resistant depression.
Capital Secured Through July 2026—With Additional Upside from Operating Revenue
More on The PennZone
NRx reports that it has secured operating capital for ongoing development through July 2026, covering the critical period leading up to the FDA's action date on the KETAFREE™ ANDA submission. With revenue now emerging from HOPE clinics and an expanding patient base, the company expects increasing non-dilutive revenue contribution.
Strategic Partnerships Strengthening Commercial Potential
NRx has partnered with Alvogen Pharmaceuticals, leveraging Alvogen's global reach to support development and commercialization of NRX-101 for suicidal bipolar depression. Additional potential indications include non-opioid chronic pain management and treatment of complicated UTIs.
A Pivotal Moment for NRXP Investors
Between its dual-path FDA strategy, real-world data advantages, strong analyst sentiment, and rapidly expanding commercial infrastructure, investors now see NRx Pharmaceuticals reaching an inflection point:
With updated clinical results, strong regulatory momentum, and multiple near-term catalysts, NRx is uniquely positioned to become a leader in next-generation therapies for suicidal depression—a market with urgent unmet need and substantial commercial potential.
For more information:
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Website: www.nrxpharma.com
Media Contact: Matthew Duffy, Chief Business Officer
Email: mduffy@nrxpharma.com | Phone: (484) 254-6134
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
The FDA submission comes as independent analyst D. Boral issues a Buy rating with a $34 price target, citing NRx's maturing regulatory position, expanding revenue-generating operations, and strong clinical catalysts across its NMDA-focused drug platform.
A Transformative Solution for a Critical Mental Health Crisis
More than 13 million Americans contemplate suicide each year, according to the CDC. Current treatment options are limited, and many lack evidence of rapid antisuicidal benefit. NRx's pipeline directly targets this gap:
NRX-100 (IV Ketamine)
- Awarded Fast Track Designation by the FDA for reducing suicidal ideation in depression, including bipolar depression.
- Supported by results from well-controlled NIH-sponsored studies as well as newly licensed data from French health authorities.
- Being pursued via an NDA, expected to be completed in Q4 2025, supported by real-world data from 60,000 IV ketamine patients versus 6,000 intranasal S-ketamine patients. Early analyses suggest faster onset and greater impact compared to nasal formulations.
NRX-101 (D-Cycloserine + Lurasidone)
- An investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression.
- New real-world evidence shows D-cycloserine may double the effectiveness of TMS, opening additional market opportunities in treatment-resistant depression and chronic pain.
KETAFREE™: A Strategic ANDA Pathway and Major Competitive Advantage
More on The PennZone
- CCHR: While Damaging Antipsychotics Win Approval, Proven Non-Drug Alternatives Remain Ignored
- Arcuri Group Announces Long‑Term Partnership with WakeMed Health & Hospitals to Deliver Situational Awareness and De‑escalation Training
- KDG Announces Acquisition of Square Foot Consultants, Expanding Business and Technology Expertise
- Colonial Nissan Service Named Top 5 Auto Repair in Feasterville-Trevose for 2025
- At 25, She Became One of the Youngest AAPI Female Founders to Win One of the World's Most Prestigious Design Awards for a Lamp That Makes You Smile
The global generic ketamine market is estimated at $750 million annually. Nearly all existing products contain benzethonium chloride (BZT)—a preservative banned from topical antiseptics and not recognized as safe by the FDA.
NRx's preservative-free KETAFREE™ directly addresses this safety concern.
The company has:
- Applied for KETAFREE™ as a proprietary product name
- Manufactured initial registration lots and prepared capacity for 1 million vials per month
- Filed a Citizen Petition urging removal of BZT from all U.S. ketamine products
- Positioned manufacturing entirely within the United States, aligning with national efforts to secure domestic drug supply chains
KETAFREE™ is strategically separate from the company's innovative NRX-100 program, enabling two simultaneous FDA pathways:
- ANDA approval (generic route) for rapid commercialization
- Fast Track NDA approval (innovative route) for suicidal depression—an unmet medical need with multibillion-dollar potential
Expanding Commercial Footprint: HOPE Therapeutics Clinics
2025 marks NRx's entry into revenue-generating operations through its HOPE Therapeutics subsidiary.
The company currently operates three clinics in Florida, with six more expected by year-end, targeting treatment-resistant psychiatric conditions, chronic pain, and military/veteran mental health.
NRx also launched Florida's first deployment of the ONE-D protocol with Ampa Health, a groundbreaking, single-day treatment approach utilizing an FDA-cleared device. Peer-reviewed results of the protocol show up to 87% response and 72% remission rates, signaling a disruptive new pathway for addressing treatment-resistant depression.
Capital Secured Through July 2026—With Additional Upside from Operating Revenue
More on The PennZone
- Juego Studios Extends Full-Cycle Game Development & Outsourcing Capabilities to the UAE Market
- VENUS Goes Live on CATEX Exchange As UK Financial Ltd Activates The Premier Division Of The Maya Meme's League
- Atlanta Tech Founder Seeks Clarity on Intellectual Property and Innovation Policy
- Purple Heart Recipient Honored by Hall of Fame Son In Viral Tribute Sparking National Conversation on Service Fatherhood, Healing and Legacy
- Maronda Homes Leverages Power of AI to Automate, Accelerate Home Warranty Claims Process
NRx reports that it has secured operating capital for ongoing development through July 2026, covering the critical period leading up to the FDA's action date on the KETAFREE™ ANDA submission. With revenue now emerging from HOPE clinics and an expanding patient base, the company expects increasing non-dilutive revenue contribution.
Strategic Partnerships Strengthening Commercial Potential
NRx has partnered with Alvogen Pharmaceuticals, leveraging Alvogen's global reach to support development and commercialization of NRX-101 for suicidal bipolar depression. Additional potential indications include non-opioid chronic pain management and treatment of complicated UTIs.
A Pivotal Moment for NRXP Investors
Between its dual-path FDA strategy, real-world data advantages, strong analyst sentiment, and rapidly expanding commercial infrastructure, investors now see NRx Pharmaceuticals reaching an inflection point:
- ✔ FDA-validated ANDA submission for KETAFREE™
- ✔ Fast Track designation for NRX-100
- ✔ Breakthrough Therapy designation for NRX-101
- ✔ $34 analyst price target from D. Boral
- ✔ Manufacturing scale ready for 1M vials/month
- ✔ Real-world ketamine data fueling NDA advancement
- ✔ Nationwide clinic expansion underway
With updated clinical results, strong regulatory momentum, and multiple near-term catalysts, NRx is uniquely positioned to become a leader in next-generation therapies for suicidal depression—a market with urgent unmet need and substantial commercial potential.
For more information:
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Website: www.nrxpharma.com
Media Contact: Matthew Duffy, Chief Business Officer
Email: mduffy@nrxpharma.com | Phone: (484) 254-6134
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
0 Comments
Latest on The PennZone
- ASTI Ignites the Space Economy: Powering SpaceX's NOVI AI Pathfinder with Breakthrough Solar Technology: Ascent Solar Technologies (N A S D A Q: ASTI)
- Hiring has reached a "Digital Stalemate"—Now, an ex-Google recruiter is giving candidates the answers
- 2026 Pre-Season Testing Confirms a Two-Tier Grid as Energy Management Defines Formula 1's New Era
- The Philadelphia Party Launches to Expand Civic Leadership, Candidate Pathways, and Education
- Platinum Car Audio LLC Focuses on Customer-Driven Vehicle Audio and Electronics Solutions
- Postmortem Pathology Expands Independent Autopsy Services in Kansas City
- Postmortem Pathology Expands Independent Autopsy Services Across Colorado
- $38 Million in U.S. Government Contract Awards Secured Through Strategic Partner. Establishing Multi-Year Defense Revenue Platform Through 2032: $BLIS
- Mecpow M1: A Safe & Affordable Laser Engraver Built for Home DIY Beginners
- CrashStory.com Launches First Colorado Crash Data Platform Built for Victims, Not Lawyers
- Inkdnylon Earns BBB Accreditation for Verified Business Integrity
- Josh Stout "The Western Project"
- Open House Momentum Builds at Heritage at South Brunswick
- A Celebration of Visibility, Voice and Excellence: The 57th NAACP Image Awards Golf Invitational, Presented by Wells Fargo, A PGD Global Production
- TopDogTours Expands Award-Winning Walking Tours in Philadelphia with Immersive Experiences
- Athens in Spring: A Culinary City Break That Rivals Paris and Copenhagen
- ClearSight Therapeutics Signs LOI with Covalent Medical for $60M Multi-Channel OTC Eye Care Partnership
- Jayne Williams Joins Century Fasteners Corp. Sales and Business Development Team
- Lord of the Rings Star Orlando Bloom To Host Special Experience at FAN EXPO Philadelphia
- Rocket Fibre Services Growing Customer Base With netElastic Networking Software